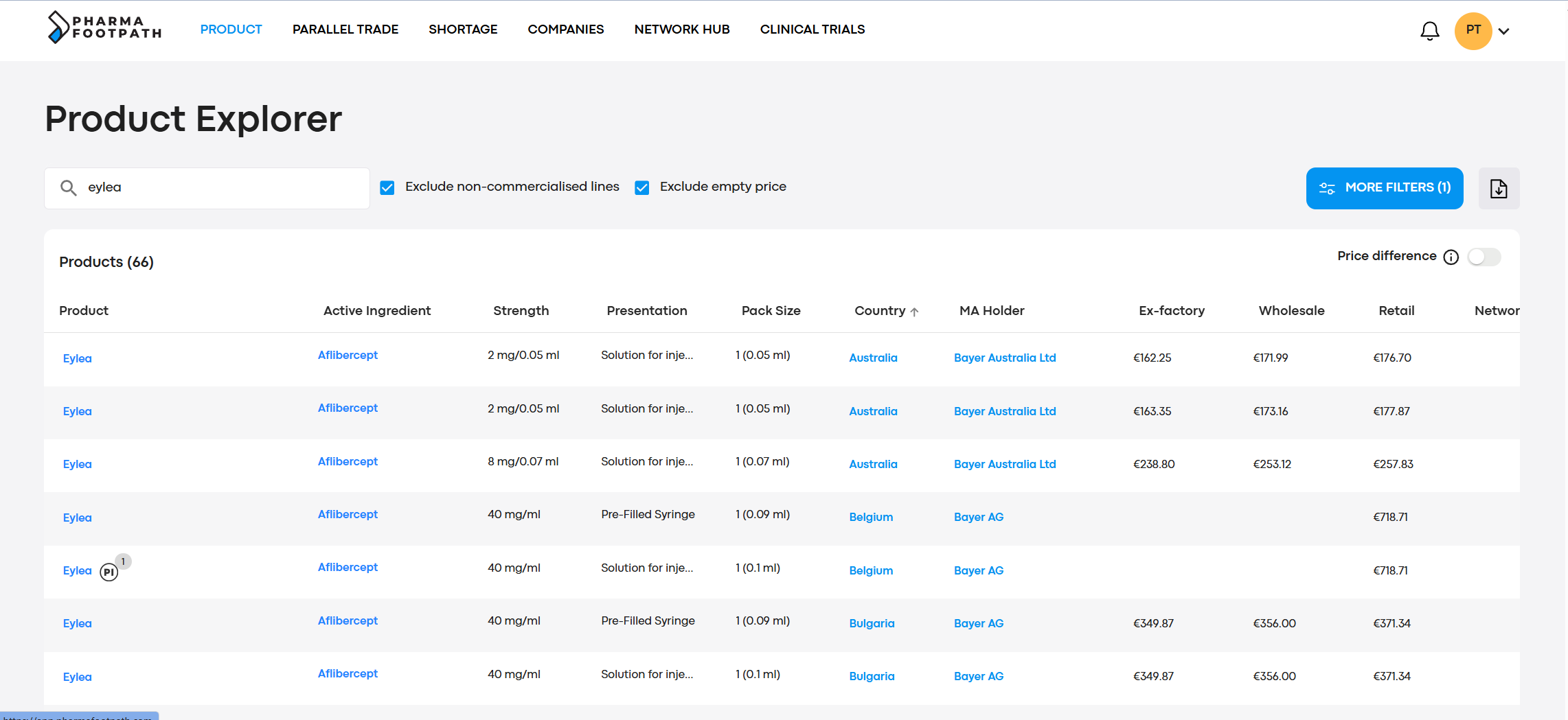
Product Explorer
Review the registration information for multiple countries in a single unified database. Filter information by MA holders, countries, presentations, strengths, pack sizes, and other characteristics.
Compare drug prices, suppliers, registrations, and shortages across global pharmaceutical markets.

PharmaFootpath unifies global drug pricing, supplier data, registrations, shortages, and clinical trial information into a single searchable platform. Instantly reveal market opportunities, empowering procurement teams, wholesalers, and parallel traders to act quickly and confidently across countries.







Pharmaceutical pricing, registrations, shortages, and supplier data are spread across national databases, regulatory agencies, and market reports. For procurement teams and pharmaceutical traders, gathering this information can take hours or days.
Fragmented landscape slows decision-making and limits visibility into international opportunities.
PharmaFootpath aggregates pharmaceutical market data from official sources and organizes it in a searchable cloud platform. Users can explore pharmaceutical markets in seconds, not hours.
Compare drug prices by country and spot new market opportunities.
Find licensed distributors and wholesalers globally.
Monitor medicine shortages and supply disruptions worldwide.
Access product registrations and parallel trade licenses.

Review the registration information for multiple countries in a single unified database. Filter information by MA holders, countries, presentations, strengths, pack sizes, and other characteristics.
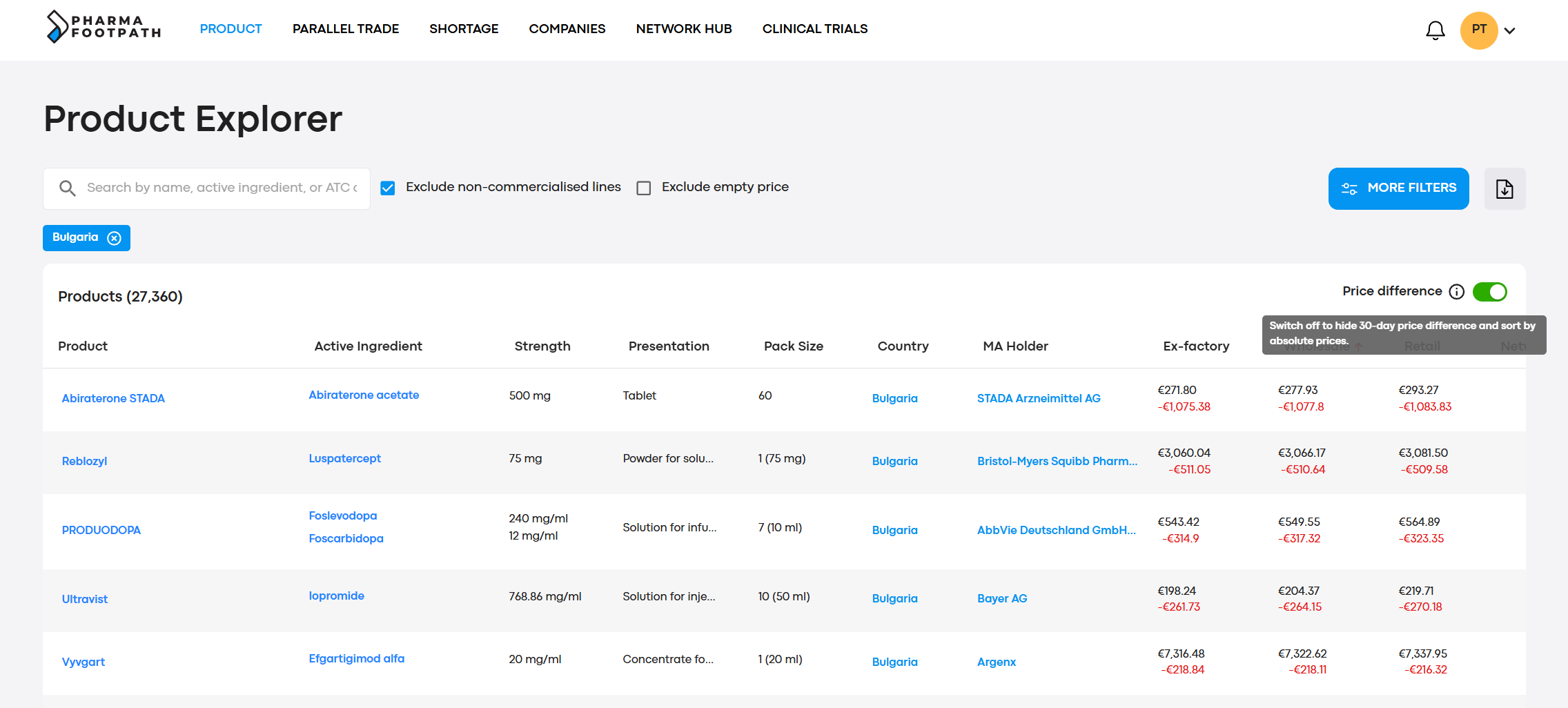
Prices are converted to the currency of your choice, with daily updated currency exchange rates. This lets you compare prices globally in a single screen, immediately.
Order information by prices, or by the largest changes in prices over the last 30 days.
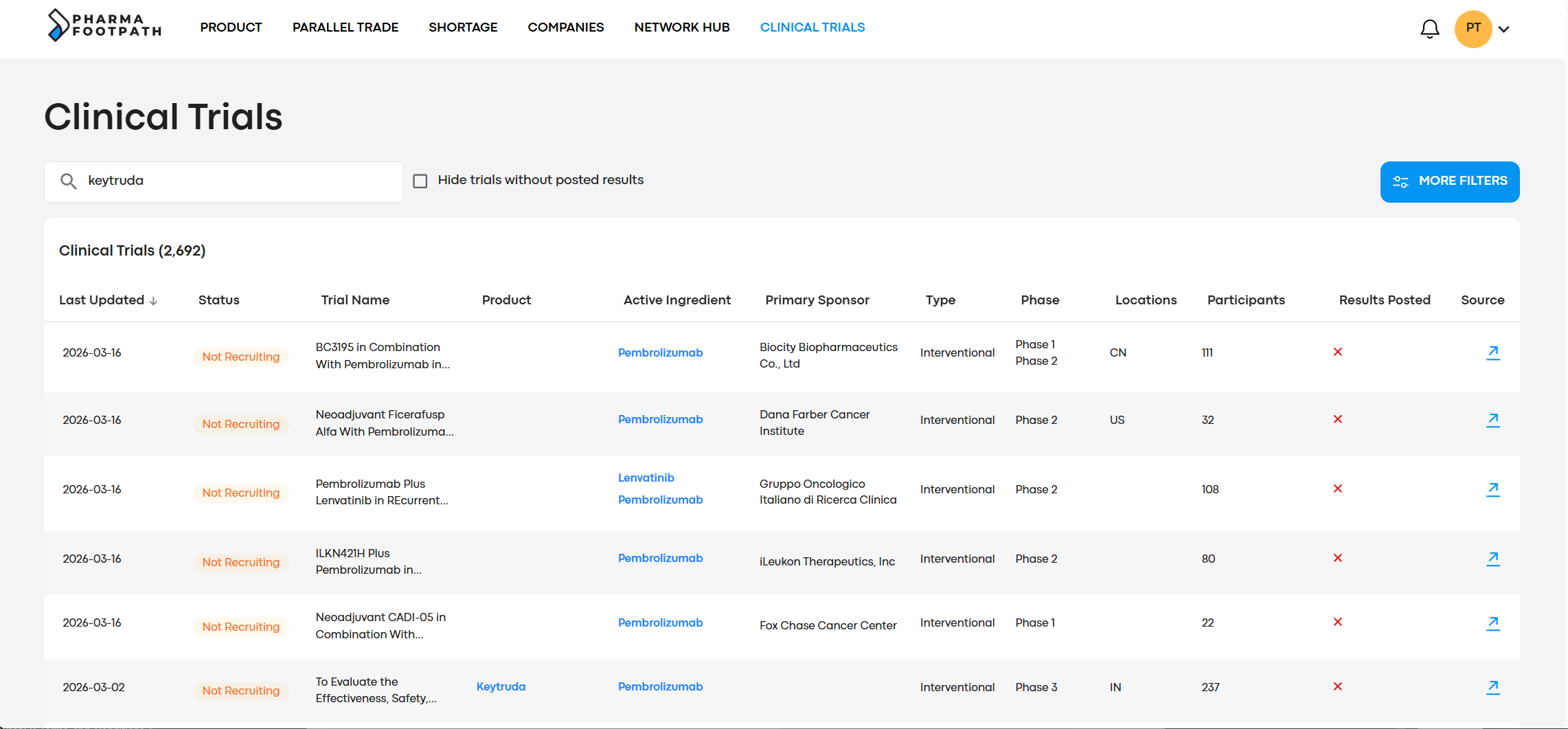
Search through multiple clinical trial registers in a single interface, connected to the drug registration data and market data of PharmaFootpath.
This helps you to select a drug name or API, and then review global clinical trials currently running with this.
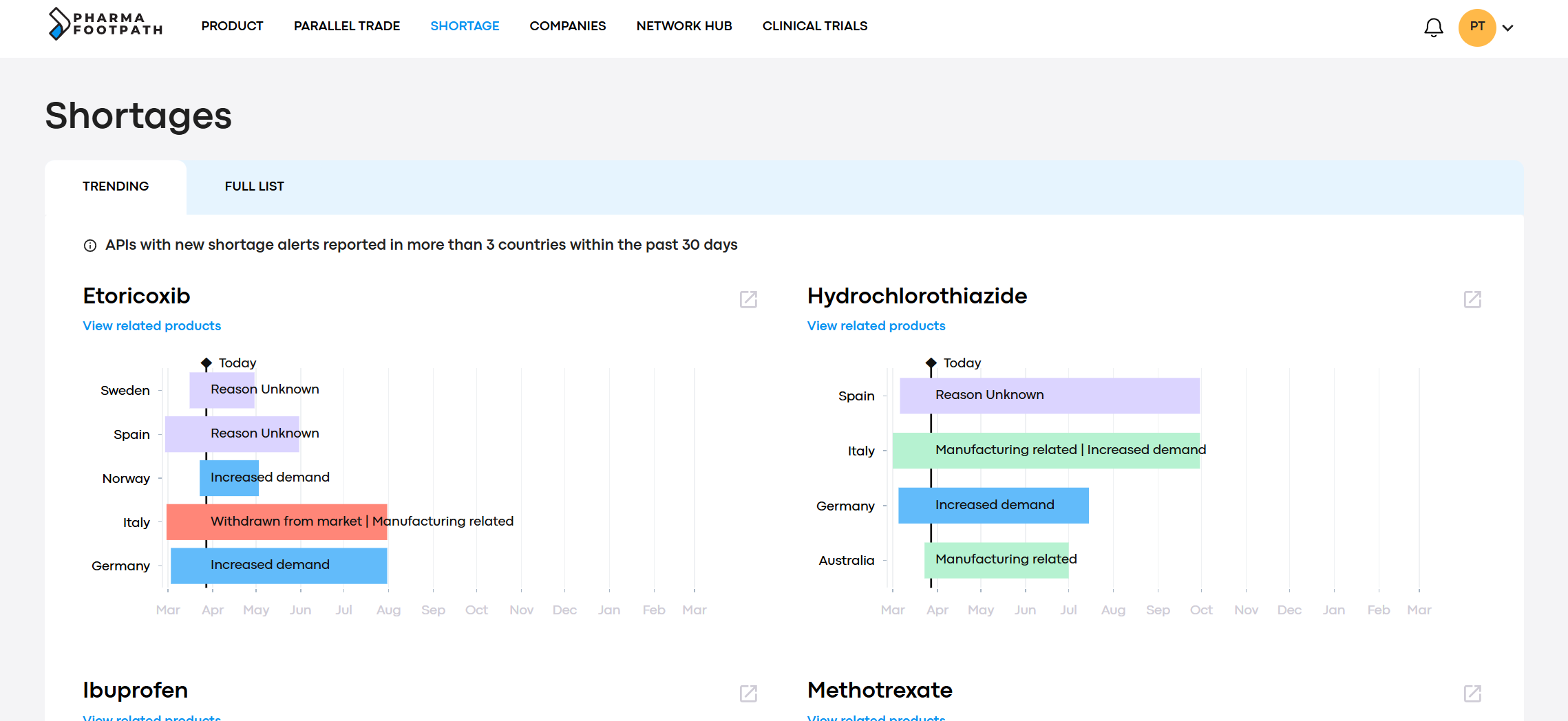
Monitoring drug shortages is an important task, but the volume of information can be difficult to manage.
PharmaFootpath collects and structures shortage information into a single interface, monitoring trends to highlight the API with which there are increasing supply disruption signals.
Procurement teams can efficiently search multiple markets, compare pricing, and identify suppliers.
Parallel traders can track product registrations, monitor competition, and find arbitrage opportunities across EU markets.
Clinical trial supply teams can identify global product availability and find new sourcing opportunities.
Pharmaceutical wholesalers can find new suppliers, partners, and international trade opportunities.
PharmaFootpath gathers pharmaceutical market data from official national organizations, regulatory databases, and verified sources. Shortage data is updated daily, and most datasets are refreshed weekly, so users have the latest information.
Explore challenges facing pharmaceutical procurement and sourcing teams in international markets.
PharmaFootpath is a pharmaceutical market intelligence platform that aggregates drug prices, supplier information, product registrations, shortages, clinical trials, and licensing data into one searchable system for pharmaceutical traders, wholesalers, and procurement teams.
PharmaFootpath is used by pharmaceutical procurement teams, parallel traders, wholesalers, and clinical trial supply companies that need visibility into pharmaceutical markets across multiple countries.
PharmaFootpath gathers pharmaceutical market data from official national organizations, regulatory agencies, and verified public sources to ensure reliable information.
Medicine shortage data is updated daily, while most other datasets, such as product registrations and pricing, are updated weekly depending on the source schedule.
PharmaFootpath focuses on European pharmaceutical markets with additional coverage for Switzerland, UK, USA, Canada, Australia, Brazil, UAE, and Saudi Arabia.
We are continually expanding our market offerings. If you’re interested in a market not listed, please contact us to check when it will be added!
We identify where products are registered and commercialised, price information, who the distributors are for the products, and various other details that accelerate sourcing products.
PharmaFootpath monitors and publishes both centrally authorised and national level parallel trade licenses in Europe. We identify the origins of national parallel trade licenses, as well as review and publish trends in the parallel trade markets.
We're focussed on providing data to support international pharmaceutical businesses. Particularly those looking to source internationally, export to new markets, identify unlicensed medicines, or considering launching portfolio products internationally.
PharmaFootpath is a cloud based application, so is accessible from any laptop or PC with internet access.

Discover new suppliers, identify trading opportunities, and compare pharmaceutical markets across countries in one platform.