Parallel Import Medicines in Europe
How Pharmaceutical Parallel Trade Works in the European Market
The European pharmaceutical market operates within a complex regulatory environment where national governments control medicine pricing, reimbursement policies, and procurement frameworks.
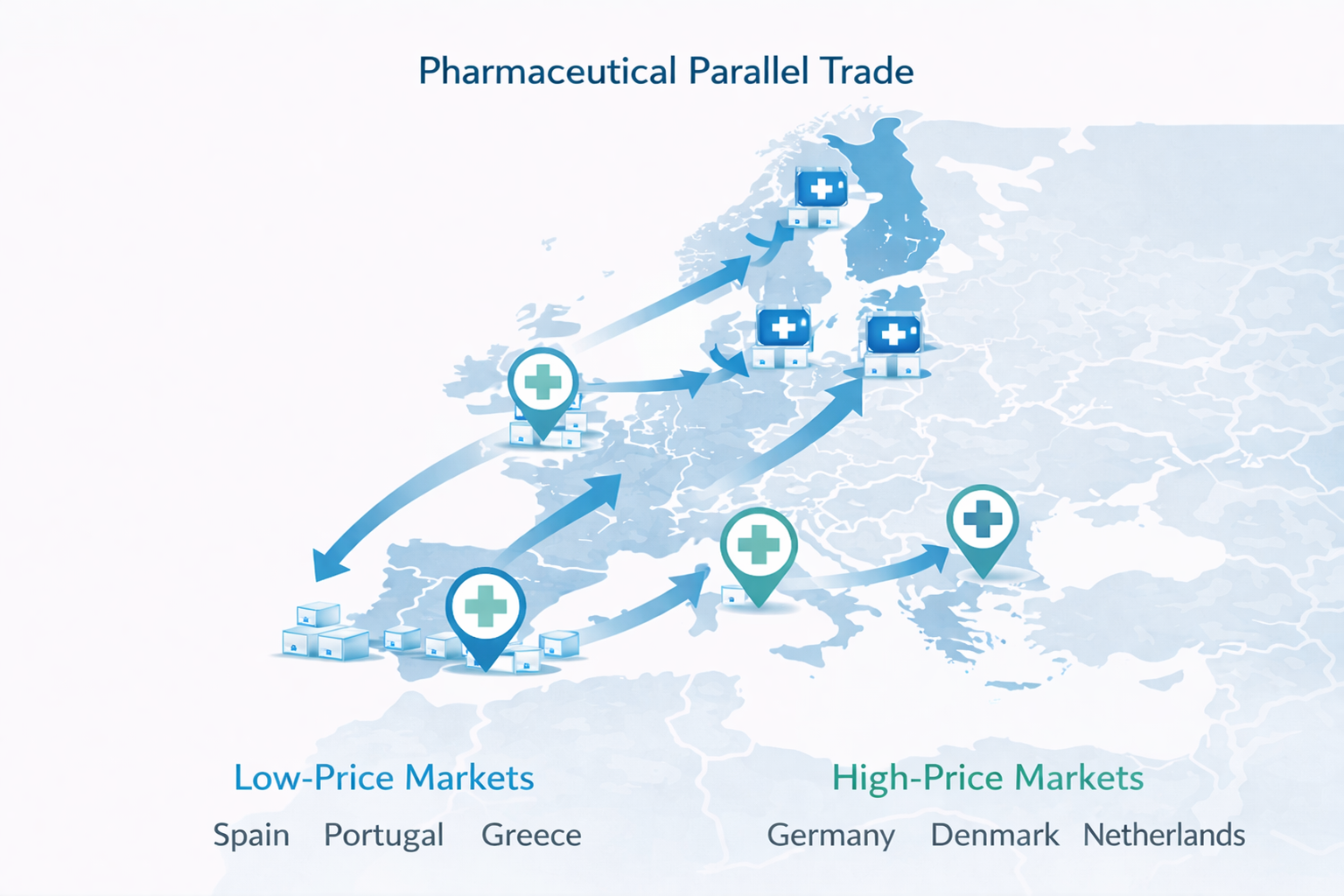
As a result, the same medicine may be sold at significantly different prices across European countries. These price differences create opportunities for distributors to purchase medicines in lower-price markets and resell them in higher-price markets through a practice known as pharmaceutical parallel trade.
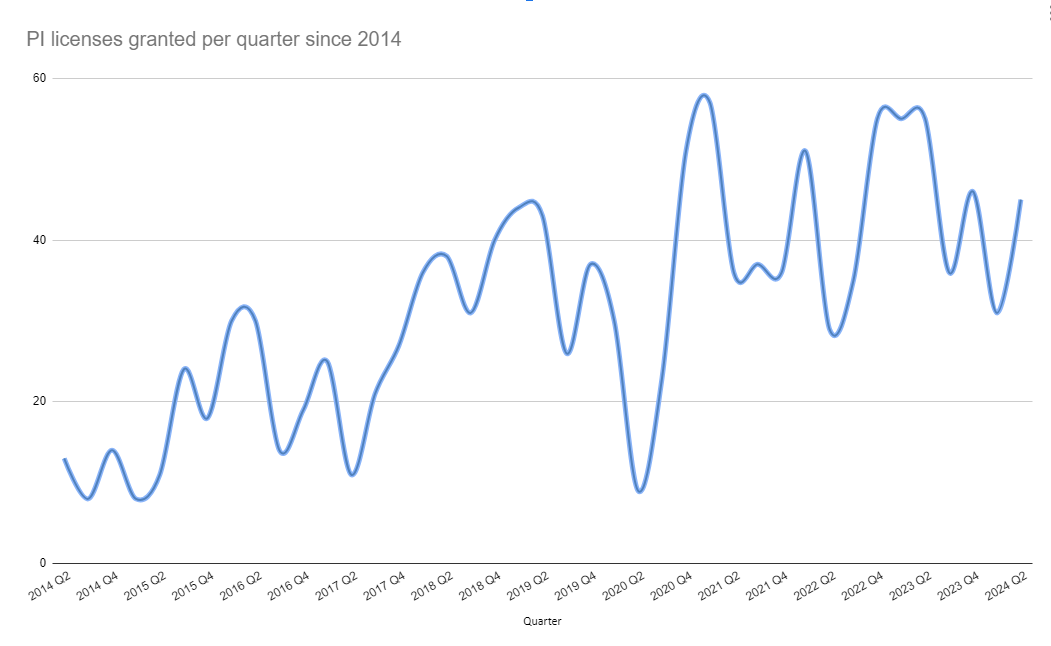
Parallel imports have become an established component of the European pharmaceutical supply chain. Today, the European parallel import market generates approximately €6–7 billion annually, representing roughly 2–3% of the European pharmaceutical market.
For pharmaceutical procurement teams, distributors, and supply-chain analysts, understanding how parallel trade works is essential for:
- identifying cross-border sourcing opportunities
- managing procurement costs
- monitoring supply chain risks
- understanding regulatory requirements.
This report explains:
- how parallel import medicines work
- the regulatory framework governing parallel trade
- the economic drivers behind pharmaceutical arbitrage
- major legal cases shaping the market
- how procurement teams identify sourcing opportunities.
What Are Parallel Import Medicines
Parallel import medicines are pharmaceutical products legally purchased in one EU or EEA country and resold in another by an independent distributor outside the manufacturer’s official distribution network.
This practice exists because medicine prices vary across Europe due to national healthcare policies and reimbursement systems.
EU law allows such cross-border trade under the principle of free movement of goods, provided the imported medicine:
- contains the same active substance
- has the same pharmaceutical form
- has a valid marketing authorisation
- meets regulatory safety and labeling requirements.
For a deeper explanation of how pharmaceutical parallel imports operate across the European supply chain, see our guide:
What Is Pharmaceutical Parallel Import? A Guide for Procurement and Supply Chain Teams
This article explains the parallel import process from sourcing and licensing through repackaging and distribution across EU markets.
European Parallel Trade Market Overview
Parallel pharmaceutical trade represents a relatively small but strategically important segment of the European medicines market.
Key market facts include:
- Parallel imports generate €6–7 billion in annual European sales.
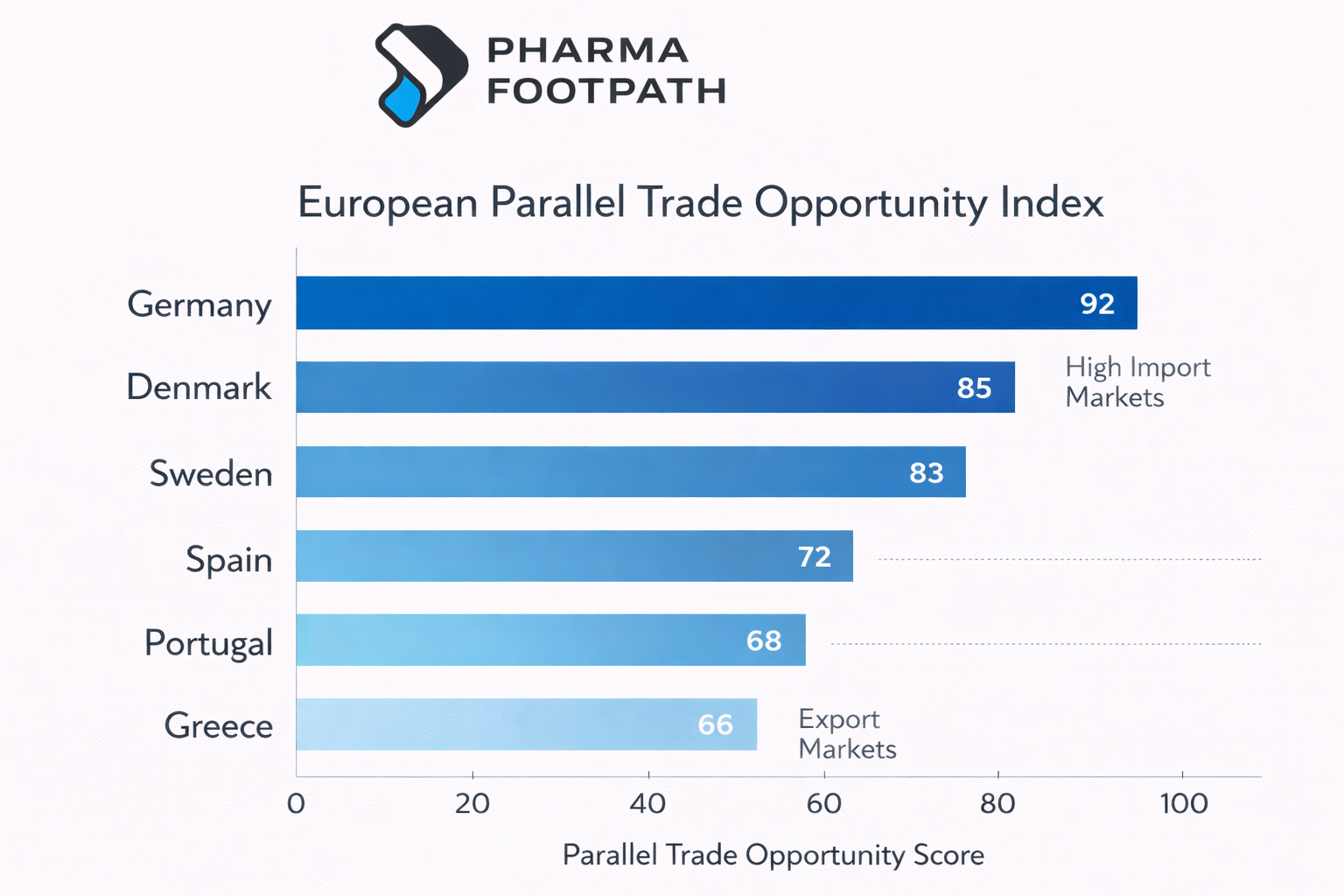
- Germany represents the largest destination market.
- Nordic countries and the Netherlands also receive significant volumes.
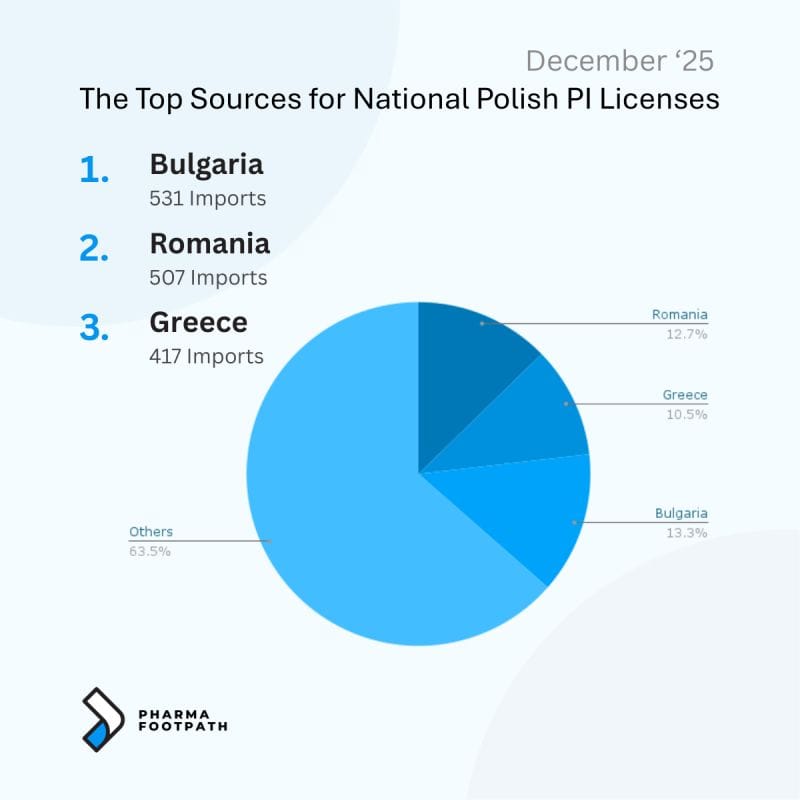
- Source markets frequently include Spain, Greece, Portugal, and Italy.
Although the overall share of the pharmaceutical market is limited, parallel trade can significantly influence procurement strategies in markets with substantial price differences between countries.
Why Parallel Imports Exist
Parallel imports exist primarily because medicine pricing is regulated nationally rather than centrally in the EU.
Each country sets pricing policies through mechanisms such as:
- reference pricing
- reimbursement negotiations
- national procurement systems
- health technology assessments.
Because these systems differ across countries, the same medicine can have substantially different prices.
Research suggests that cross-country price differences can exceed 20–40% for some medicines, creating economic incentives for distributors to engage in parallel trade.
Economic Drivers of Pharmaceutical Parallel Trade
Several structural factors drive parallel pharmaceutical trade across Europe.
National Pricing Systems
Each EU country independently regulates medicine prices and reimbursement policies.
Free Movement of Goods
The EU single market allows authorised products to move freely between Member States.
Distributor Arbitrage
Specialised wholesalers purchase medicines in lower-price countries and resell them in higher-price markets.
Procurement Demand
Hospitals and pharmacies often seek alternative supply sources to reduce procurement costs.
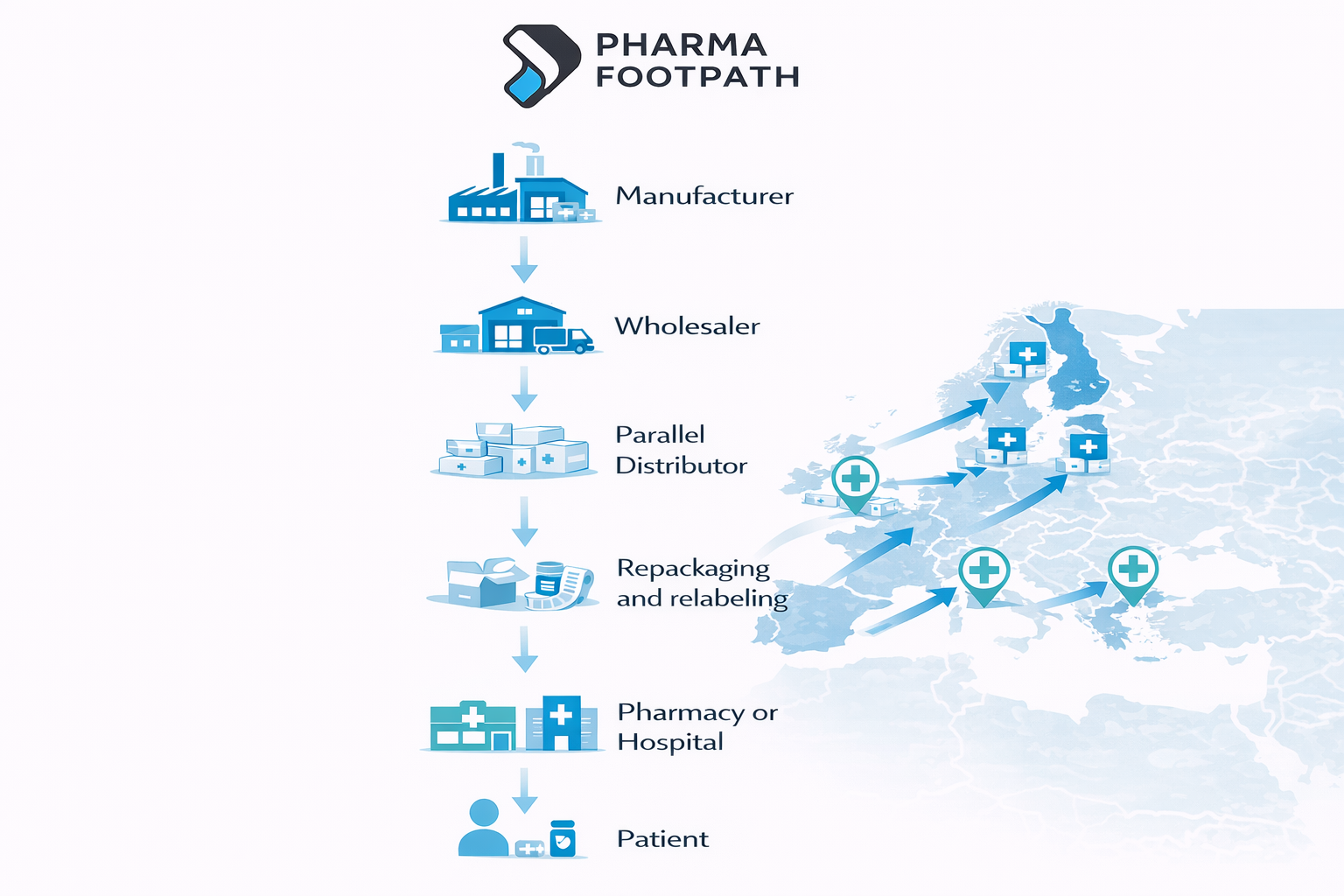
How Parallel Import Works in the Pharmaceutical Supply Chain
Parallel imports involve several operational stages.
1. Procurement in Lower-Price Markets
Parallel traders identify medicines with significant price differences between EU markets.
Products are purchased from:
- pharmaceutical wholesalers
- authorised distributors
- secondary supply networks.
Example:
A distributor may purchase oncology medicines in Spain and resell them in Germany.
2. Repackaging and Relabeling
Imported medicines often require packaging modifications to comply with national regulations.
This may include:
- translating patient leaflets
- adding regulatory labeling
- adjusting packaging formats.
3. Distribution into Higher-Price Markets
After regulatory approval and packaging adaptation, medicines may be distributed to:
- pharmacies
- hospitals
- national procurement agencies
- wholesalers.
Because the imported medicine is therapeutically identical to the authorised product, it can typically be dispensed under existing medical regulations.
Parallel Import vs Parallel Distribution
Parallel import and parallel distribution are related but distinct concepts.
Regulatory Framework for Parallel Imports
Parallel trade operates under several European legal principles.
These include:
Parallel trade is permitted because once a product has been placed on the market within the EU by the manufacturer or with its consent, it may generally be redistributed across Member States.
Because parallel trade operates through national licensing systems, companies often need to track where their medicines are being imported and redistributed.
A detailed explanation of this process can be found in:
How to Identify Parallel Trade Licenses in the EU: A Guide for MA Holders
The EU parallel trade ecosystem includes more than 95,000 parallel trade licenses, issued by national regulators across European markets.
Key Court Cases That Shaped Pharmaceutical Parallel Trade
Several landmark decisions by the Court of Justice of the European Union (CJEU) have shaped how parallel pharmaceutical trade operates.
Bristol-Myers Squibb v Paranova (1996)
One of the most influential cases in pharmaceutical parallel trade is Bristol-Myers Squibb v Paranova.
The Court established five conditions under which parallel importers may repackage medicines without violating trademark rights.
These conditions include:
- Repackaging must be necessary to market the product in the importing country.
- The product must remain unaltered in its original condition.
- The packaging must clearly identify both the manufacturer and the importer.
- The repackaged product must not damage the trademark's reputation.
- The importer must notify the trademark owner before marketing the product.
If these conditions are not satisfied, manufacturers may oppose the sale of repackaged medicines.
GlaxoSmithKline v European Commission (2009)
Another major case involved GlaxoSmithKline's attempt to restrict exports of medicines from Spain to higher-price markets such as the United Kingdom.
The Court ruled that agreements designed to restrict parallel trade could violate EU competition law because they artificially partition the internal market.
This decision reinforced the EU’s commitment to maintaining free movement of goods within the internal market.
Examples of Parallel Import Medicines
Parallel trade frequently occurs in therapeutic areas with high medicine prices.
These examples illustrate how distributors exploit cross-border price differences to supply medicines to higher-price markets. Parallel trade dynamics also vary significantly by country.
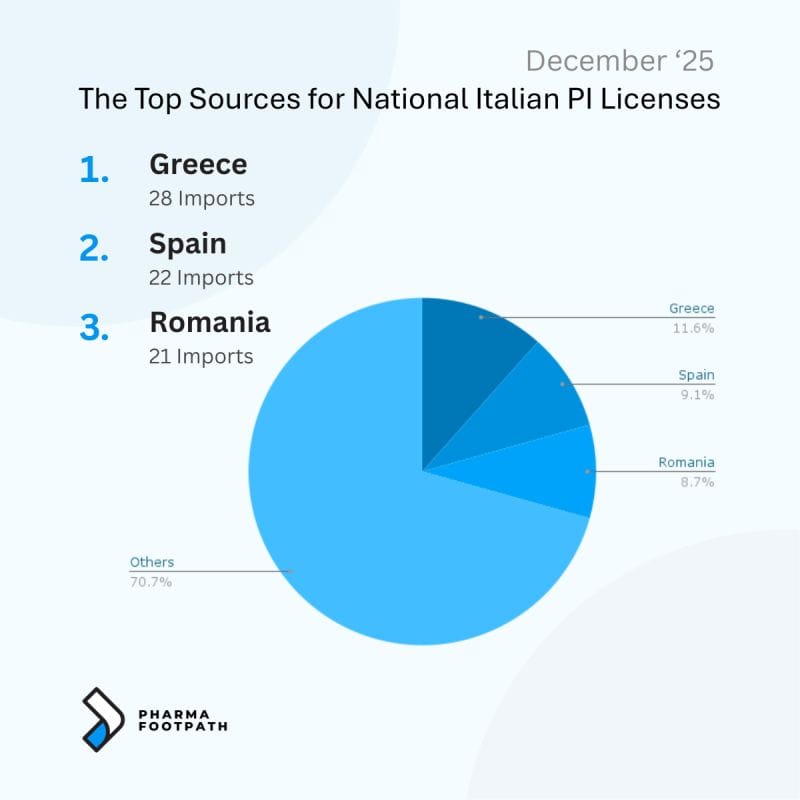
For example, the Italian market currently has over 1,400 products, with companies competing to secure supply opportunities.
A deeper analysis of the country-specific dynamics is available in:
Italian Parallel Import Market
Benefits of Parallel Import Medicines
Parallel trade can generate several benefits for healthcare systems.
Lower Healthcare Costs
Parallel imports increase price competition and can reduce medicine costs.
Supply Chain Flexibility
Additional supply channels may help address temporary shortages.
Market Competition
Manufacturers may adjust pricing strategies in response to parallel trade activity.
Risks and Challenges
Parallel trade also presents challenges.
Medicine Shortages
Large-scale exports may reduce the availability of medicines in lower-priced countries.
Regulatory Complexity
Parallel importers must comply with strict licensing, pharmacovigilance, and serialization requirements.
Manufacturer Resistance
Pharmaceutical companies sometimes attempt to limit parallel trade through supply restrictions.
How Procurement Teams Identify Parallel Trade Opportunities
Pharmaceutical procurement teams increasingly rely on market intelligence to identify cross-border sourcing opportunities.
Key indicators include:
- medicine price differences between countries
- issuance of parallel trade licenses
- distributor activity across markets
- medicine shortage signals.
However, this information is often fragmented across regulators, wholesalers, and supply chain databases.
Market intelligence platforms such as PharmaFootpath help integrate these datasets.
They allow procurement teams to analyze:
- cross-country price differences
- distributor networks
- regulatory approvals
- supply chain disruptions.
Learn how our parallel trade intelligence platform helps pharmaceutical procurement teams monitor cross-border supply opportunities.
Monitoring Parallel Trade with Market Intelligence Platforms
Tracking parallel trade requires access to multiple datasets across European pharmaceutical markets.
Key intelligence sources include:
- regulatory licensing databases
- medicine pricing data
- distributor networks
- shortage monitoring systems.
Platforms such as PharmaFootpath combine these data sources into a unified intelligence platform that helps pharmaceutical professionals monitor supply chain dynamics across Europe.
Discover Parallel Trade Opportunities Across Europe
Access pricing intelligence, distributor networks, and regulatory approvals across European pharmaceutical markets.
Parallel Trade Opportunity Index
European Parallel Trade Opportunity Index
Data Sources for Parallel Trade Analysis
Parallel trade analysis typically combines multiple public and commercial datasets.
These include:
- national medicine price databases
- parallel trade licensing registers
- pharmaceutical distributor networks
- medicine shortage reporting systems.
Platforms such as PharmaFootpath aggregate these datasets to provide pharmaceutical professionals with structured intelligence on cross-border medicine trade.
Future Outlook for Pharmaceutical Parallel Trade
Parallel trade is expected to remain a structural feature of the European pharmaceutical market.
As long as national pricing systems create differences between countries, distributors will continue to identify cross-border sourcing opportunities.
At the same time, regulators must balance:
- cost reduction
- supply stability
- pharmaceutical innovation.
Increasingly, procurement teams rely on market intelligence platforms to monitor supply chain dynamics across European markets.
Key Takeaways
Parallel import medicines play a significant role in the European pharmaceutical supply chain.
They:
- redistribute medicines across markets with different prices
- create procurement opportunities for distributors
- increase competition in healthcare markets
- introduce both economic benefits and supply chain risks.
Understanding these dynamics is essential for pharmaceutical professionals operating across European markets.
Try PharmaFootpath Intelligence Parallel Trade Market Platform
FAQ: Pharmaceutical Parallel Import
What is pharmaceutical parallel trade?
Pharmaceutical parallel trade involves buying medicines cheaply in one European country and reselling them at higher prices in another, relying on price disparities and the EU's free movement of goods.
Are parallel-imported medicines safe?
Yes. Parallel-imported medicines are already authorised medicines that must meet the same regulatory safety, quality, and pharmacovigilance requirements as locally supplied medicines.
Why do medicine prices differ across Europe?
Medicine prices differ because each European country regulates pharmaceutical pricing and reimbursement independently through national healthcare policies.
What is the difference between parallel imports and parallel distribution?
Parallel imports involve medicines authorised through national regulatory procedures, while parallel distribution refers to centrally authorised medicines moved between EU countries under the supervision of the European Medicines Agency.
Is parallel pharmaceutical trade legal?
Yes. Parallel trade is legal in the European Union under the principle of free movement of goods, provided that the imported medicine is therapeutically equivalent to the product authorised in the destination market.
What role do data platforms play in parallel trade analysis?
Procurement teams increasingly use pharmaceutical market intelligence platforms to monitor medicine prices, distributor activity, regulatory approvals, and shortage signals across markets, identifying cross-border sourcing opportunities.
About the Author

Phil Thomas has had a varied background, including tech product management and international trade of pharmaceuticals. While primarily in clinical trial supply, he supported companies in unlicensed medicines, parallel trade, drug shortages, and managed access programmes.
He has previously sold a big data business and served as managing director at a cybersecurity consultancy. He has a large collection of unread books, which he promises to read one day.